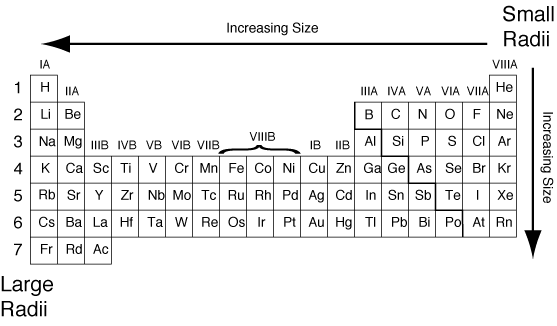
  Courtesy of The Comprehensive Nuclear-Test-Ban Treaty (CTBT) photo stream.įrom there the United States and the Soviet Union carried out a further series of open-air tests of atomic weapons.  In general, the size of an atom will decrease as you move from left to the right of a certain period.The Ivy Mike thermonuclear test, November 1, 1952. How does atomic radius increase?Īn atom gets larger as the number of electronic shells increase therefore the radius of atoms increases as you go down a certain group in the periodic table of elements. Atomic radius decreases as you move across a period from left to right and decreases as you move up a group from bottom to top. This is due to trends in the periodic table, and the effective nuclear charge that holds the valence electrons close to the nucleus. Across a period, effective nuclear charge increases as electron shielding remains constant. In general, atomic radius decreases across a period and increases down a group. What is atomic radius on the periodic table?Ītomic radius is the distance from the atom’s nucleus to the outer edge of the electron cloud. What is the order of the elements from smallest to largest atomic radius?Īnswer and Explanation: With these information, we can now list the elements in increasing atomic radius as O< C < S n < S r. Therefore, Na has the largest atomic radius. Again, Cl lies to the right and Na lies to the left of the periodic table. In periodic table, 3rd period will have larger atomic radius than the second period. Which element in period three has the largest atomic radius?Īnd Cl : belong to the 3rd period. Which ion below has the largest radius *?īecause K+ has the greatest nuclear charge (Z = 19), its radius is smallest, and S2− with Z = 16 has the largest radius.  Which element has the largest atomic radius in group 18? Which atom has the largest atomic radius K GA BR RB? Silicon has the largest atomic number and therefore silicon has the smallest atomic radius because atomic radii decrease from left to right across a period. Which of the following atoms has the largest and which has the smallest radius explain your answer in terms of trends in the periodic table?Įxplain your answer in terms of trends in the periodic table. Here, Mg + 2 will have largest ionic radius. Ionic radius increases with increase in positive charge on isoelectronic species. Which of the following has largest radius a Mg 2 +) B Na (+) C O 2 -) d/f (-)? Has highest radii because by going left to right in the periodic table, the size get decreased so. Which has the largest radius Co3+ Mn3+ fe3+ Cr3+? Plane mirror has the largest radius of curvature. Which of the following has larger radius of curvature? Rubidium is the second most reactive metal and is very soft, with a silvery-white lustre. Rubidium (Rb), chemical element of Group 1 (Ia) in the periodic table, the alkali metal group. How many atoms does RB have?Ģ more rows What is RB on the periodic table? As you can see from the diagrams, the same atom could be found to have a different radius depending on what was around it. The radius of an atom can only be found by measuring the distance between the nuclei of two touching atoms, and then halving that distance. So Cesium has the largest atomic radius among the given elements. Trends in the atomic radius showing that moving down the periodic table, size increases, whereas moving across the periodic table from left to right, size decreases. Has the largest atomic radius whereas helium has the smallest. Which atom has the largest atomic radius quizlet? The carbon atom has the greater atomic size among all the other choices they are nitrogen, fluorine, and oxygen. The one proton has a more major consequence than one electron thus, the electrons are drawn towards the atomic nucleus, causing a smaller radius. The Periodic Table: Atomic Radius, Ionization Energy, and Electronegativityġ8.0 similar questions has been found Which has largest atomic size? Atomic Radius – Basic Introduction – Periodic Table Trends, Chemistry 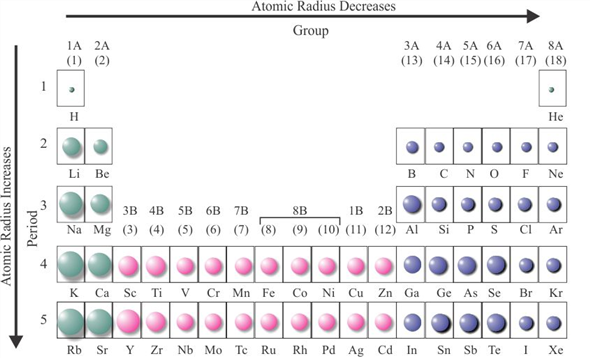 Which of the following has largest radius? Its atomic radius is 0.248 nm and the (+1) ionic radius is 0.148 nm. The electronic configuration of Rubidium is (Kr)(5s 1). Which has a larger atomic radius Rb or SR?Īmong the given elements, Rb has the largest atomic radius as the atomic radius increases down the group and decreases across the period. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
